What is dalton's atomic theory3/30/2024 The law of multiple proportions states that if two elements form more than one compound between them, the masses of one element combined with a fixed mass of the second element form in ratios of small integers. Of course, we just as easily could have illustrated the law by considering the mass of nitrogen that combines with one gram of oxygen it works both ways!.Note that just as the law of multiple proportions says, the weight of oxygen that combines with unit weight of nitrogen work out to small integers there is a typo in Line 3-where there is a 3, there should be a 4. Line 3 is obtained by dividing the figures of the Line 2 by the smallest O:N ratio in Line 2 (which is the ratio for N 2O).Dalton proposed that all matter is made of tiny indivisible atoms, which he imagined as. See all videos for this article By far Dalton’s most influential work in chemistry was his atomic theory. The laws of conservation of mass and definite proportions can be explained using the concept of atoms. Atomic theory of John Dalton See how John Dalton built his atomic theory on principles laid out by Henry Cavendish and Joseph-Louis Proust John Dalton and the development of the atomic theory. All matter is composed of atoms that are indivisible. But someone who depends solely on experiment would work these out by finding the mass of O that combines with unit mass (1 g) of nitrogen. Dalton made a few assumptions in his atomic theory 1-4. The numbers in Line 2 are just the mass ratios of O:N, found by dividing the corresponding ratios in line 1. 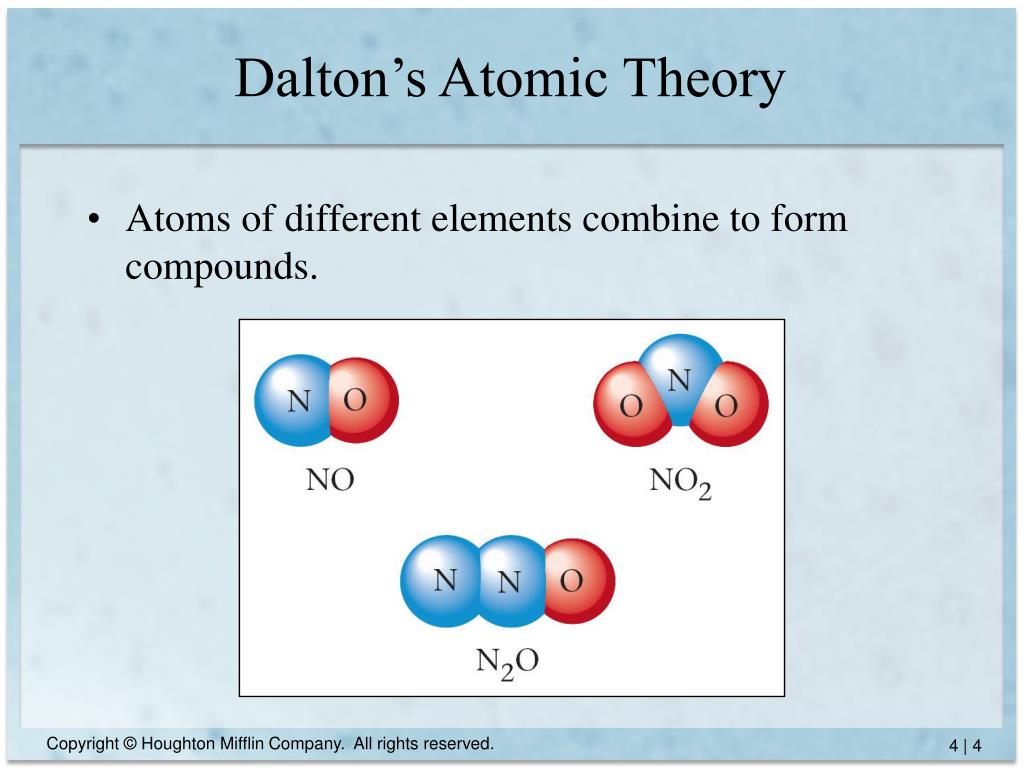
(These numbers were not known in the early days of Chemistry because atomic weights of most elements were not reliably known.) Scientists have taken the hypothesis of Greek scholars and have built on it with their different discoveries and theories regarding the atom, which derives from the Greek word 'atomos,' meaning indivisible. 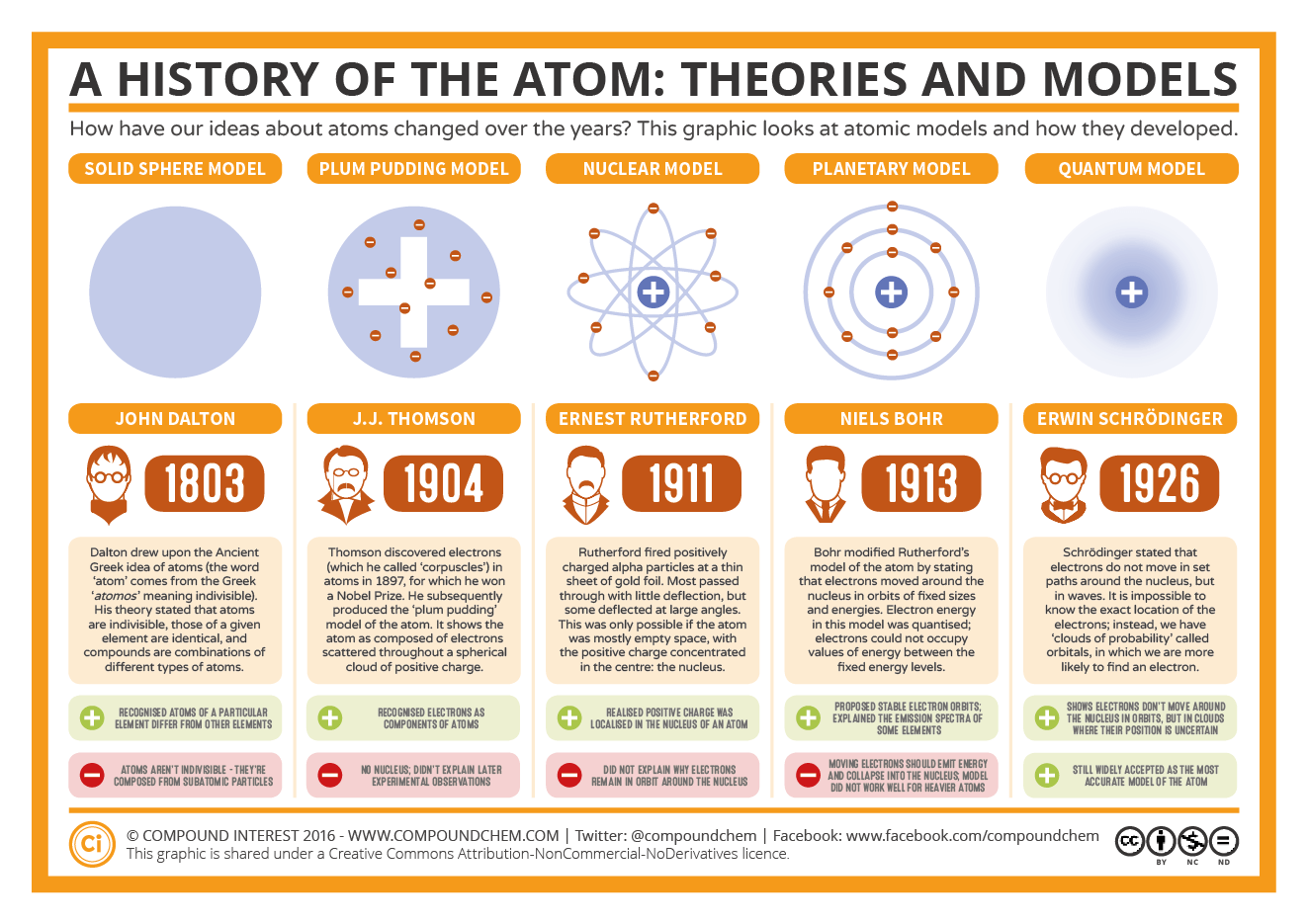
However, it was later established that atoms of the same element can have different masses. Dalton’s Atomic Theory: In the course of this research into gases, Dalton also discovered that certain gases could only be combined in certain proportions, even if two different compounds shared. Thus for NO 2, we have (1 × 14) : (2 × 16) = 14:32. Atomic theory has evolved since ancient times. Dalton states that atoms of a given element have precisely the same masses. These ratios were calculated by simply taking the molar mass of each element, and multiplying by the number of atoms of that element per mole of the compound. His findings were based on experiments and the laws of chemical combination. Line 1 shows the ratio of the relative weights of the two elements in each compound. John Dalton, a British school teacher, published his theory about atoms in 1808. 
However, he made a mistake in assuming that the simplest compound of two elements must be binary, formed from atoms of each element in a 1:1 ratio, and his system of atomic weights was not very accurate - he gave oxygen an atomic weight of seven instead of eight.ĭespite these errors, Dalton's theory provided a logical explanation of concepts, and led the way into new fields of experimentation.\)) compounds. 
Using his theory, Dalton rationalised the various laws of chemical combination which were in existence at that time. The theory proposed a number of basic ideas:Īll atoms of the same element are identicalĭifferent elements have different types of atomsĬhemical reactions occur when atoms are rearrangedĬompounds are formed from atoms of the constituent elements. He stated his theory in a lecture to the Royal Institution in 1803. Dalton's theory was based on the premise that the atoms of different elements could be distinguished by differences in their weights.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
